Home » Office for Research Protections » Radioisotopes » Radioisotopes Events, Training, & Resources » Transitioning Active UIC Authorizations into CATS Safety
UIC Menu
ORP Menu
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
Transitioning Active UIC Authorizations into CATS Safety
In January 2026, all active UIC authorizations will migrate to CATS Safety; these migrated protocols contain essential information from existing authorizations. The CATS Safety platform supports a single lab-based protocol under each Principal Investigator, so researchers with multiple authorizations will find their authorizations have been combined into one protocol. Teaching and Facility protocols will remain separate.
All Principal Investigators with active UIC authorizations must complete the full transition of their radiation safety research into CATS Safety by accessing the migrated protocol in CATS Safety and answering all required protocol questions when either of these two scenarios occurs:
- Amendment – To change a currently approved authorization.
- Three-Year Renewal (De Novo Renewal) – De Novo submissions in CATS Safety must be completed a minimum of 2 weeks before your protocol expiration date.
Please refer to the instructions below for guidance on completing this transition. Live training opportunities are also available. See the CATS Safety Guidance page for more information on using the system.
How to transition active UIC authorizations via Amendment
- Log in to CATS Safety
Go to https://researchsafety.psu.edu. - Access Submissions
From the left-hand menu, click Submissions.
- Locate Active Protocols
In the Submissions workspace, select the Active tab. - Select the Protocol
Review the list and click the Name of the protocol associated with the PI who needs to create an Amendment.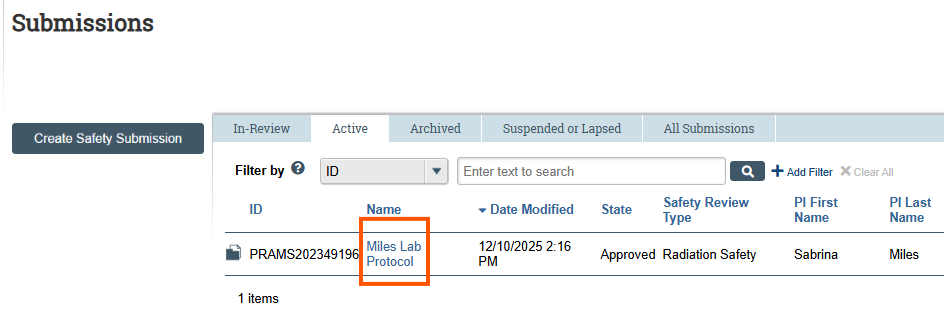
- Accessing Currently Approved Authorizations
In the protocol workspace, go to the Documents tab. Under Protocol documents are currently approved UIC authorizations for this protocol. Refer to these documents if/as needed to complete the CATS Safety submission.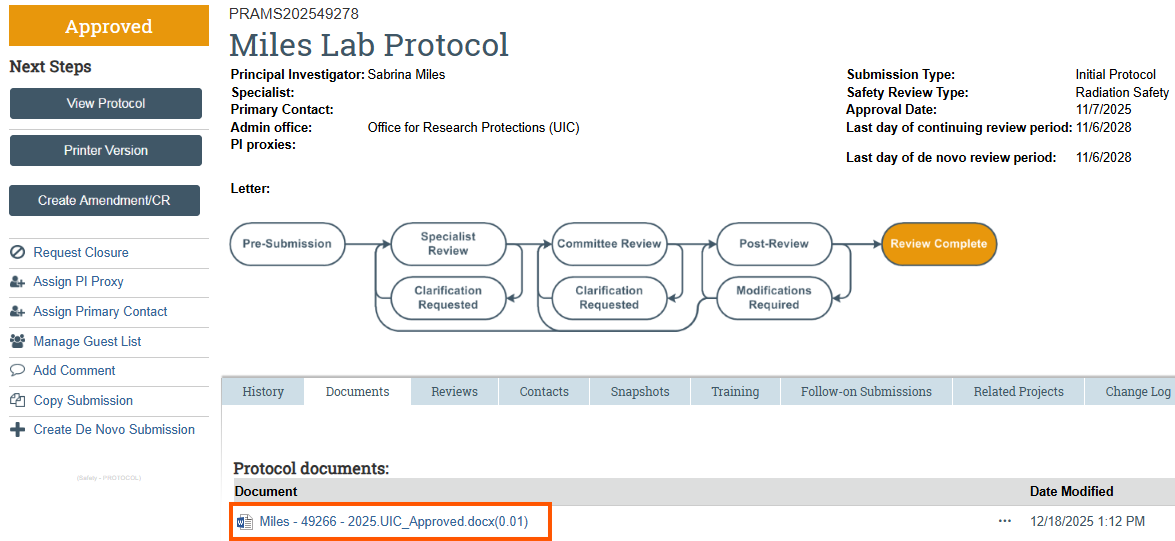
- Create Amendment
From the left-hand menu under Next Steps, click Create Amendment/CR. - Choose Submission Type
On the Amendment/Continuing Review page, select Amendment as the Type of Submission, then click Continue. - Complete Amendment Introduction
Answer all required questions on the Amendment Introduction page and click Continue.
Refer to Radiation Safety Protocol Tips document for guidance. - Update Protocol Details
Review and complete each page of the protocol, clicking Continue to move forward. - Save & Exit
On the final page (Supporting Documents), click Save & Exit. - Submit for Review
Note: The submission is not yet submitted for review. The PI must click the Submit activity from the protocol workspace to forward the submission for review.
How to transition active UIC authorizations via De Novo Submission
- Log in to CATS Safety
Go to https://researchsafety.psu.edu. - Access Submissions
From the left-hand menu, click Submissions.
- Locate Active Protocols
In the Submissions workspace, select the Active tab. - Select the Protocol
Review the list and click the Name of the protocol associated with the PI who needs to create a De Novo submission.
- Accessing Currently Approved Authorizations
In the protocol workspace, go to the Documents tab. Under Protocol documents are currently approved UIC authorizations for this protocol. Refer to these documents if/as needed to complete the CATS Safety submission.
- Create De Novo Submission
From the left-hand menu under Next Steps, click Create De Novo Submission.
- Confirm Creation
A confirmation window will appear. Select Yes and click OK to proceed. - Monitor Creation Process
CATS Safety will begin creating the submission. On the History tab of the protocol, you will see a prompt to refresh the browser. Continue refreshing until the De Novo submission is created. - Locate the New Submission
Once created, a hyperlink to the De Novo submission will appear on the History tab. The new submission ID will match the original protocol ID with the prefix DN1 (indicating the first De Novo submission).
- Open the De Novo Submission
Click the hyperlink to access the new submission. - Edit Protocol
Click Edit Protocol to begin updating the submission.
- Complete Protocol Pages
Review and complete each page of the protocol, clicking Continue to move forward. - Save & Exit
On the final page (Supporting Documents), click Save & Exit. - Submit for Review
Note: The submission is not yet submitted for review. The PI must click the Submit activity from the protocol workspace to forward the submission for review.