Home » Office for Research Protections » Research Integrity » Respondent Guidance
Research Integrity Menu
ORP Menu
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
Respondent Guidance

A Respondent is the person against whom an allegation of research misconduct is directed or who is the subject Research misconduct is defined as fabrication, falsification, and plagiarism (FFP) in proposing, performing, or reviewing research, or in reporting research results. Research misconduct does not include honest error or scientific differences of opinion.
If you are a Respondent, use the information on this page to guide you through the process. Refer to Policy RP02 Addressing Allegations of Research Misconduct for additional information, including an expanded definition, roles, and the lifecycle of misconduct proceedings.
Who's Involved and Confidentiality
- The Research Integrity Officer (RIO) limits the disclosure of any allegations and protects the identities of those involved, although complete anonymity cannot be guaranteed. You will receive key notifications, copies of interview transcripts, and copies of Inquiry and Investigation reports for comment .
- The Complainant (the person making the allegation of research misconduct) may be interviewed as a witness and will receive notifications about the outcome of the Inquiry and Investigation, but does not direct the research misconduct process or receive copies of the Inquiry or Investigation reports.
- College and department and leadership may be involved on a need to know basis (e.g., if data is sequestered).
- Sponsors and relevant University administrators will also be notified of the outcome pursuant to RP02 and/or any relevant sponsor-specific regulations or policies.
Your Responsibilities
- Take this important opportunity to participate in the process through full cooperation. This will allow for consistent application of the policy, opportunities for both the person bringing forward the concern and the person being accused to provide information, documentation, and testimony of events and ensures a rigorous process for evaluating potential research integrity issues.
- Written records are more helpful than verbal recollections.
- Forward original emails with a brief summary of relevance.
- Specify where and how research data is stored (specific locations, devices, file formats, etc.).
- Identify relevant types and sources of evidence.
- Provide any documentation that may be relevant and helpful to the review.
- You may be interviewed during the Inquiry stage. If the process advances to the Investigation stage, RP02 requires that both the Complainant and Respondent be interviewed.
- Proceedings will continue regardless of your participation.
- A lack of cooperation or efforts to delay will be documented as part of the case record.
- Respondents may raise conflicts of interest as defined in RP02 with faculty selected to assist with the case; you have 10 days to object proposed committee members or Officials for both Inquiry or Investigation.
- Conflicts may include unresolved personal, professional, or financial issues.
- Faculty are vetted for expertise and impartiality; outside experts may be also be consulted when more specialized expertise is needed.
- The RIO decides whether a conflict or lack of expertise disqualifies a faculty member.
- Respond to the preliminary Reports; after the process concludes you will no longer have an opportunity to do so.
- Focus comments on the relevant science and include only necessary details.
- Provide supporting evidence when possible/appropriate.
- Direct comments to the Inquiry Official or Committee; they will review them before finalizing the reports. This is your opportunity to correct inaccuracies, raise issues, and/or respond to preliminary determinations made by the Official or Committee.
- The Deciding Official reviews both the full report and Respondent’s comments before making a final decision.
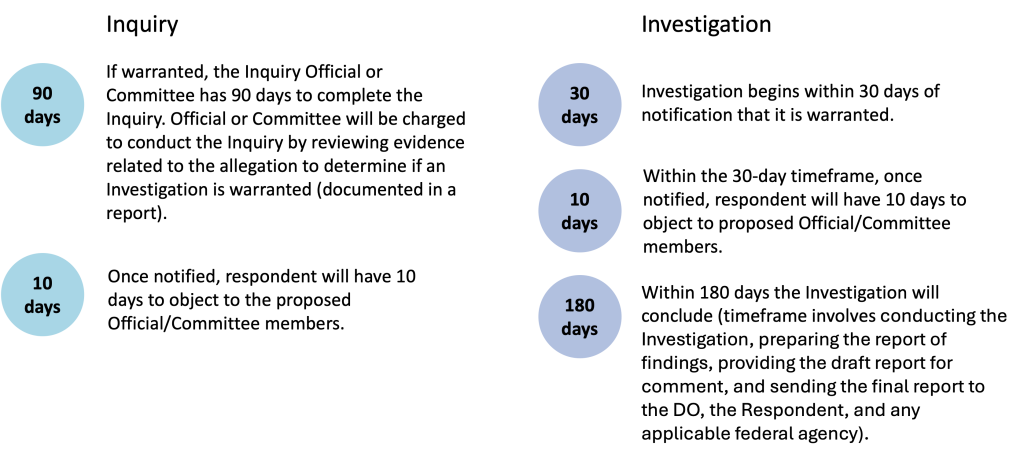
Extended Description for Timeline
If warranted, the Inquiry Official or Committee has 90 days to complete the Inquiry. Official or Committee will be charged to conduct the Inquiry by reviewing evidence related to the allegation to determine if an Investigation is warranted (documented in a report).
Once notified, the Respondent will have 10 days to object to the proposed Official/Committee members.
Within the 30-day timeframe, once notified, the Respondent will have 10 days to object to proposed Official/Committee members.
Investigation begins within 30 days of notification that it is warranted.
Within 180 days the Investigation will conclude (timeframe involves conducting the Investigation, preparing the report of findings, providing the draft report for comment, and sending the final report to the DO, the Respondent, and any applicable federal agency).
What Happens Next
University Response to Allegations
- In responding to an allegation (performing an Assessment), the University will provide the following to the Respondent:
- notification of receipt of an allegation if moving to Inquiry,
- confidentiality will be maintained to the fullest extent possible,
- a copy of the preliminary Inquiry Report for comment,
- notification whether an Investigation will be initiated, and
- a copy of the preliminary Investigation Report for comment.
- Allegations that fall within the definition of research misconduct and are specific and credible enough to identify potential evidence must be reviewed regardless of the Complainant’s motives; bad faith allegations will be referred to the Office of Ethics and Compliance and may be addressed under Policy AD67.
- Allegations may be updated throughout the process to add/remove concerns or Respondents; if so, you will be notified.
- The next steps beyond the Allegation stage are available on our webpage.
Continuation of Research Activities
The RIO aims to avoid delays in the Respondent’s research. Data sequestration may briefly pause work (usually no more than 3 days). Research may continue, but Respondents should not pursue publication or funding for the work under review without consulting the RIO. Interim actions, including pausing the research, may be taken if necessary (see RP02 Section IV).
Expanded Support for Graduate Students, Faculty and Staff
We recognize this may be a difficult and stressful time, and want you to know that a variety of resources are available to support you through this process.
Graduate Students
College Level
Graduate students may reach out to the following for support and assistance:
- Department Head
- Graduate Program
- Administrator for Graduate Education in their College or School
- Associate Dean for Graduate Student Affairs of the Graduate School
Student Affairs
Student Affairs can help you continue with your studies and participation in the Penn State Community while your case is being investigated and decided.
Support and Assistance Provided:
- Understand what to expect in a transparent, open manner
- Engage with academic accommodations and University resources that can support you
- Connect with community resources that may have no ties to Penn State
- Develop self-supporting strategies to help you pursue other goals
- Explain the responsibilities and options that you have while going through the process
- Support you as you choose a response in keeping with your best interest
Advisers
Graduate students may consult with their adviser throughout the research misconduct process, except in certain situations.
When to Consult:
- Adviser does not have a direct role in the research misconduct case
When NOT to Consult:
- When adviser is the person bringing the complaint
- A research misconduct allegation has been made against you and your adviser
Ombudspersons
Support and Assistance Provided:
- Safe environment to discuss issues and concerns
- Assist with identifying or creating options for resolution
- Help understand relevant policies, connect with resources for support and assistance
- Make referrals to formal channels with investigatory powers
Ombudspersons Do NOT:
- Formally advocate for the student or any individual view (but do promote a fair process for all)
- Keep formal records
- Perform formal investigations
- Advocate for either one party or another
Faculty and Staff Respondent Support
RP02 Addressing Allegations of Research Misconduct
Penn State’s Policy RP02 process is an academic process and not a legal process and meetings and interviews are not legal proceedings. RP02 provides that a Respondent (the person against whom an allegation of research misconduct has been made) “may consult with legal counsel or a non-lawyer personal adviser (who is not a principal or witness in the case) to seek advice.” Examples of non-lawyer personal advisers include (but are not limited to) a; partner/spouse, friend, family member, ombudsperson, and/or professional or personal mentor who is not also a lawyer. Non-legal support persons cannot be a University employee unless they are currently an ombudsperson. Although uncommon, if the inclusion of a support person is desired, a request should be made in writing to the Research Integrity Officer (RIO) at least 5 business days in advance of any meeting or interview.
Department Heads, Research Deans, Supervisory Officials
For most cases, the department head and associate research dean, and/or appropriate supervisory official of the Respondent are notified of the research misconduct allegation(s) so the the department head, associate research dean, and/or supervisory official can:
- Assist the RIO with the coordination of data sequestration
- Act as support for the Respondent, as they will know the circumstances of the allegation
- Provide clarification about the research misconduct process to the Respondent
Employee Assistance Program (EAP)
Penn State’s EAP offers resources to full-time, benefits-eligible employees, their spouse and dependents, as well as members of their household. Services include (but are not limited to) in-the-moment support, short-term counseling, legal consultation and financial expertise, all of which are confidential.
Faculty Ombudspersons
The Faculty Ombudsperson is available to consult with Tenure-Line and Non-Tenure-Line full time faculty. Faculty Ombudspersons advocate for fairness.
Support and Assistance:
- Provide a safe environment for the discussion of issues and concerns
- Assist faculty in identifying or creating options for resolution that meet the needs of the faculty and the university, including referrals to formal channels with investigatory powers
Ombudspersons Do NOT:
- Keep formal records
- Mediate
- Perform formal investigations
- Advocate for either one party or another
Academic Unit Ombudspersons
In addition to the Faculty Ombudsperson, Penn State Policy AC76 requires each college and campus to have a unit ombudsperson.
- Locate the appropriate college and unit ombudsperson
- Some colleges and units may have also established additional ombudspersons who are available to assist students and/or staff as well as faculty
Findings and Corrective Actions
In order to make a finding of research misconduct, an Investigation Committee must find by a preponderance of evidence that:
- Research misconduct occurred (as defined in RP02 and/or applicable federal regulations);
- The behavior alleged to be research misconduct is a significant departure from accepted practices of the relevant research community; and
- Respondent committed the research misconduct intentionally, knowingly, or recklessly.
If a finding of research misconduct is substantiated, the University will take relevant actions as outlined in RP02. The University determines the appropriate corrective actions, not the Complainant; the Complainant will generally not receive information regarding proposed or ordered corrective actions. Sponsors and relevant University administrators will be notified of the outcome of these proceedings pursuant to RP02 and any relevant sponsor-specific regulations or policies.
Even if there is not a finding of research misconduct, there may still be a need for corrective actions. Potential corrective actions which may result from a substantiated finding of research misconduct include:
- Corrections to the scientific record (retractions, erratum, corrigendum, statements, etc.)
- Retraining/re-education
- Monitoring/proactive review of future research/submissions
- Removal from the project(s) in question
- Letter of reprimand in file
- Probation, suspension, or termination
- Revocation of degree
The imposition of corrective actions by the University does not prevent additional corrective actions from being taken by relevant sponsor/other oversight agencies.
Questions
If you have questions or concerns, please reach out to researchconcerns@psu.edu.
Although you are able to reach out to anyone on our team directly, it’s best to include researchconcerns@psu.edu on all communication to avoid delays.
Office for Research Protections
Address
200 Innovation Blvd.
Suite 110
University Park, PA 16802
The Office for Research Protections (ORP) ensures that research at the University is conducted in accordance with federal, state, and local regulations and guidelines that protect human participants, animals, students, and personnel involved with research.