Home » Office for Research Protections » IACUC » CATS IACUC Basic Steps » Respond to Clarifications
IACUC Menu
- Guideline to Report Animal Welfare Concerns
- Report an Adverse Event or Non-Compliance Incident
- IACUC Program Home
- CATS IACUC
- Policies, Guidelines, & SOPS
- AAALAC Accreditation
- Committee Information
- IACUC Meeting Dates
- Events, Training and Resources
- CATS IACUC Basic Steps
- IACUC Review Process
- Post-Approval Quality Assurance (APAQA)
- Guideline to Report Animal Welfare Concerns
- Report an Adverse Event or Non-Compliance Incident
- IACUC Program Home
- CATS IACUC
- Policies, Guidelines, & SOPS
- AAALAC Accreditation
- Committee Information
- IACUC Meeting Dates
- Events, Training and Resources
- CATS IACUC Basic Steps
- IACUC Review Process
- Post-Approval Quality Assurance (APAQA)
ORP Menu
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
Respond to Clarifications
Once a protocol has been created and submitted to the IACUC, the IACUC staff and/or IACUC committee may have additional clarifications or required modifications that need to be made by the research team before a determination can be made. As a result, the research staff may need to respond to questions and/or make changes to their protocol.
When clarifications/required modification requests are made, an email will be sent to the PI, Proxy, and Primary Contact and the submission will also appear in My Inbox within CATS IACUC.
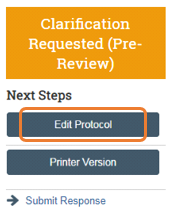
From the submission workspace History Log, click the Clarifications Request… activity entry and read the comments. Click Edit Protocol to view where reviewer notes have been added in the IACUC submission form, as well as to make changes to the submission.
The left navigation bar will identify smartform pages where reviewer notes have been added. Reviewer notes are identifiable by the reviewer note icon (![]() ).
).
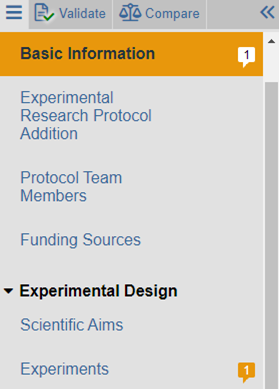
In the example above, one reviewer note was added on the Basic Information page and one reviewer note was added on the Experiments page. To read the specific reviewer note, first navigate to the page on which the reviewer note was added. Once on the page, click on each reviewer note icon (They can appear at the top right of the page (if applicable to the entire page) or to the right of any question. If applicable, reply to the reviewer note by clicking Reply.
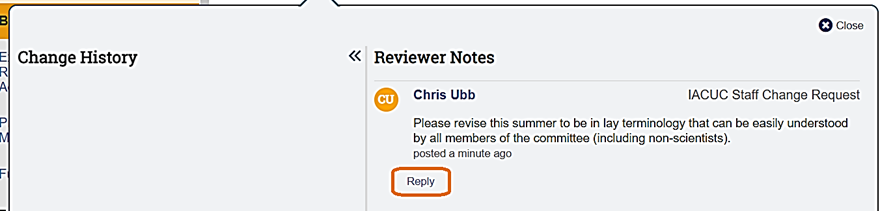
Upon clicking Reply, you will be provided with the opportunity to add text and/or attach files (if applicable). After adding any desired text and files, click OK to save the note and then Close on the Reviewer Note window.

Once all reviewer notes have been addressed and the appropriate changes have been made to the submission form, click Save and then Exit at the bottom right.
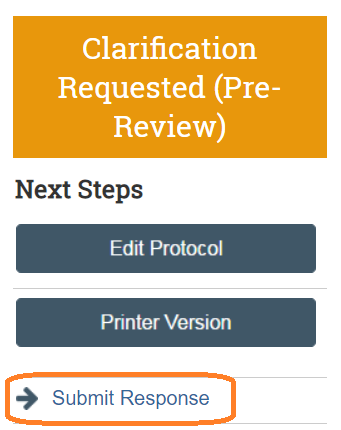
The Principal Investigator or, if assigned, a PI Proxy may now click execute the Submit Response activity in the when ready to submit back to the IACUC.
Office for Research Protections
Address
200 Innovation Blvd.
Suite 110
University Park, PA 16802
The Office for Research Protections (ORP) ensures that research at the University is conducted in accordance with federal, state, and local regulations and guidelines that protect human participants, animals, students, and personnel involved with research.