Home » Office for Research Protections » IACUC » CATS IACUC Basic Steps » Post-Approval Actions
IACUC Menu
- Guideline to Report Animal Welfare Concerns
- Report an Adverse Event or Non-Compliance Incident
- IACUC Program Home
- CATS IACUC
- Policies, Guidelines, & SOPS
- AAALAC Accreditation
- Committee Information
- IACUC Meeting Dates
- Events, Training and Resources
- CATS IACUC Basic Steps
- IACUC Review Process
- Post-Approval Quality Assurance (APAQA)
- Guideline to Report Animal Welfare Concerns
- Report an Adverse Event or Non-Compliance Incident
- IACUC Program Home
- CATS IACUC
- Policies, Guidelines, & SOPS
- AAALAC Accreditation
- Committee Information
- IACUC Meeting Dates
- Events, Training and Resources
- CATS IACUC Basic Steps
- IACUC Review Process
- Post-Approval Quality Assurance (APAQA)
ORP Menu
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
- Office for Research Protections (ORP) Home Page
- Conflict of Interest
- Controlled Substances
- Drone Operations
- Dual Use Research of Concern (IRE)
- Education
- Export Control
- Human Stem Cells (ESCRO)
- Human Subjects Research (IRB)
- Radioisotopes (UIC)
- Institutional Biosafety Committee (IBC)
- Research Data Management Policy
- Research Integrity
- Research Security
- Scientific Diving
- Vertebrate Animal Care and Use (IACUC)
- Quality Assurance
- ORP Team
- ORP Events
- ORP Announcements
Post-Approval Actions
Approved submissions can be found from My Inbox by selecting the “IACUC” tab and then “Submissions.”
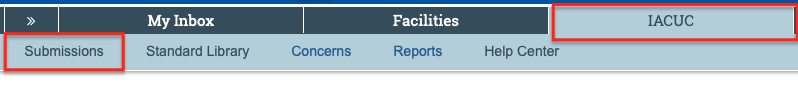
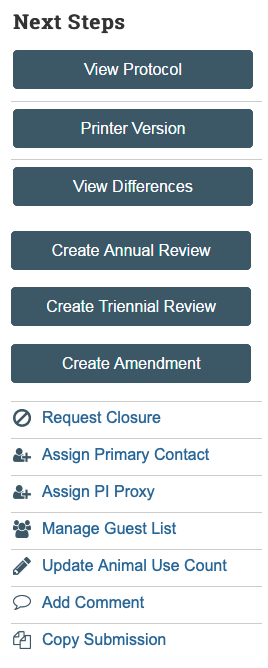
After a study has been approved, researchers may need to submit or be asked for “follow-on submissions.” Follow-on submissions is a term that is used when referencing an amendment, annual review or triennial review. The PI is responsible for annual reviews, triennial reviews, and creating any necessary amendments. All of these options are listed under the “Next Steps” menu (see picture below) for the approved protocol, which can be found in the parent protocol workspace.
When adding personnel through a modification or amendment, make sure to assign them to procedures on the Procedure Personnel Assignment page as well.
Office for Research Protections
Address
200 Innovation Blvd.
Suite 110
University Park, PA 16802
The Office for Research Protections (ORP) ensures that research at the University is conducted in accordance with federal, state, and local regulations and guidelines that protect human participants, animals, students, and personnel involved with research.